Services
At Chemometrica, we provide specialized technical consulting and professional training in chemometrics and analytical chemistry, applied to data-driven process development.
Our services are designed to support the implementation of Quality by Design (QBD) and Process Analytical Technology (PAT) principles, in alignment with the ICH guidelines, ensuring analytical robustness, regulatory compliance, and process understanding.
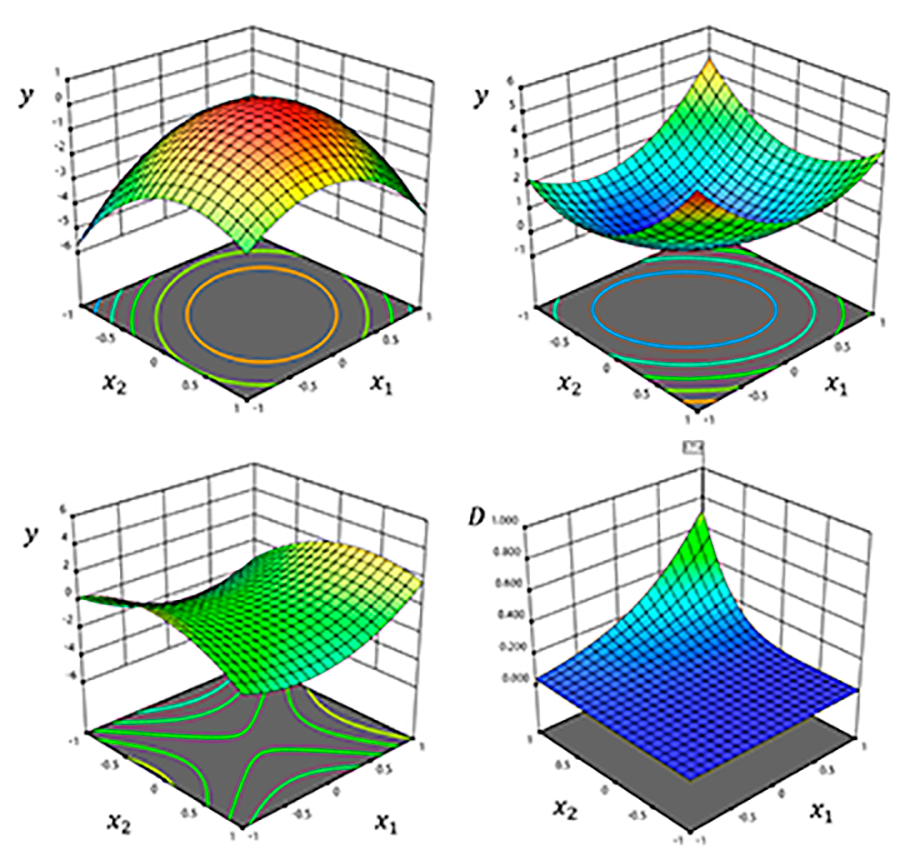
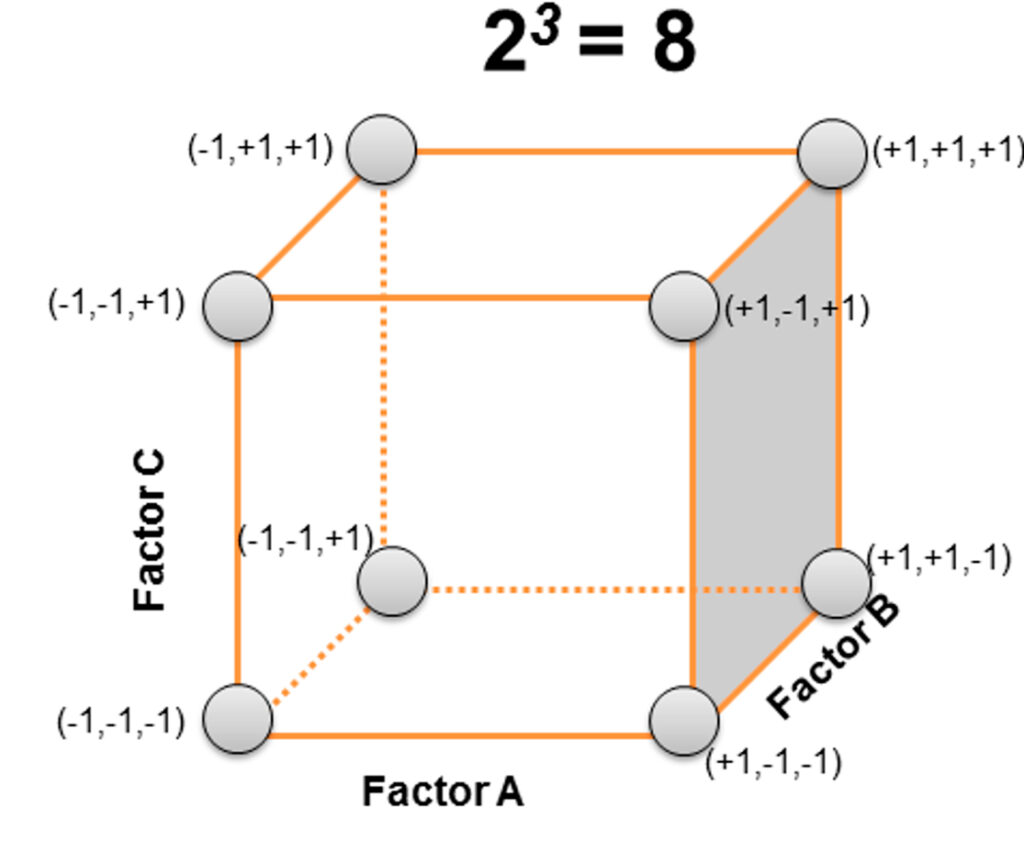
Design of Experiments (DoE)
Design of Experiments enables the efficient planning and optimization of unit operations and analytical methods through the development of statistically sound models, while minimizing experimental effort. This approach allows the identification of Critical Process Parameters (CPPs) and their relationship with Critical Quality Attributes (CQAs) — key concepts within the QbD framework — thereby improving process knowledge and product quality.
We assist our clients in the application of DoE methodologies, providing guidance in experimental design, model building, and statistical interpretation.
Multivariate Calibration
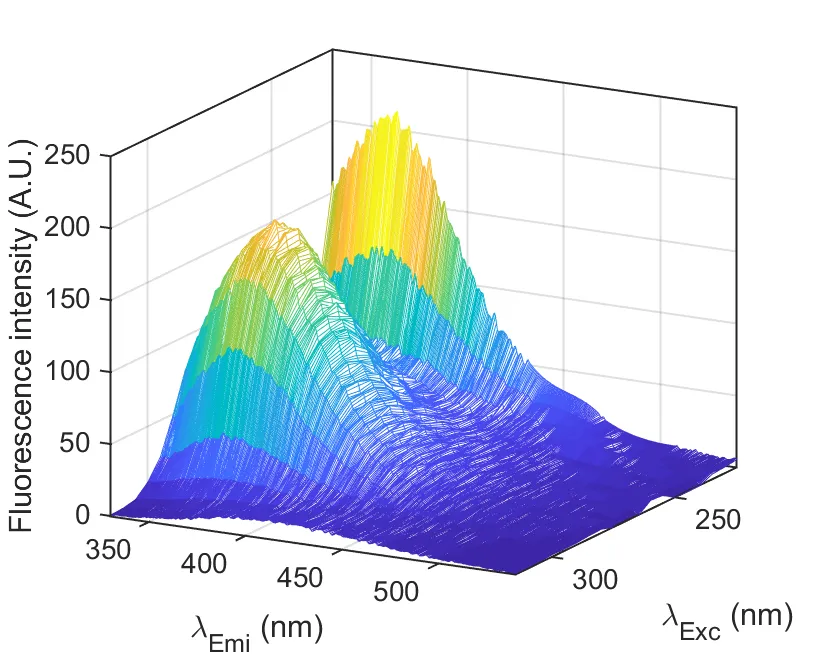
Multivariate calibration provides a powerful framework for developing analytical methods capable of monitoring and controlling quality in complex systems. Unlike conventional univariate approaches, multivariate models integrate the full spectral or sensor information, improving analytical performance in terms of accuracy, precision, and sensitivity. These methods rely on clean, safe, non-destructive instrumental techniques requiring minimal sample preparation, and are well suited for automation and real- ime monitoring under the PAT framework.
We support our clients in developing, validating, and implementing multivariate models for quantitative analysis and process monitoring.
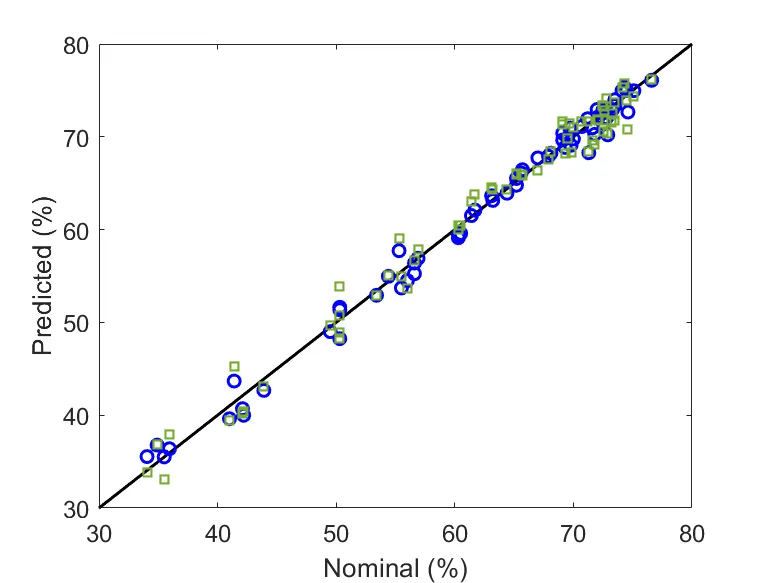
Analytical Method Validation
Analytical method validation ensures that an analytical procedure is suitable for its intended purpose, establishing reliability and compliance with regulatory expectations. A sound validation strategy combines experimental design, statistical evaluation, and proper data handling to verify method accuracy, precision, linearity, and robustness.
We guide our clients through the design, execution, and statistical assessment of validation protocols, ensuring scientifically sound and regulatory- compliant outcomes.

Training and Capacity Building
At Chemometrica, we do not provide “black box” solutions.
We believe that meaningful results emerge from collaboration and understanding. For that reason, we actively involve our clients in each stage of the analytical and modeling process, fostering a transparent and personalized experience.
In line with this philosophy, training represents a key component of our services. We offer flexible, fully remote programs—ranging from full-length courses to short seminars—covering topics such as multivariate calibration, method validation, experimental design, and applied chemometrics.
Our team has a strong academic background and extensive teaching experience, and we truly enjoy sharing knowledge. Each program combines theoretical foundations with hands-on case studies and includes custom-designed learning materials tailored to the client’s analytical and process context.
One of our main goals is to raise awareness within R&D and analytical teams about the value and benefits of adopting chemometric-based approaches, helping organizations build a common language that enhances collaboration and maximizes the impact of our consulting work.
We are Chemometrics Innovation Future
All services are tailored to the specific needs of each client or technology adopter, ensuring
alignment with their analytical objectives, regulatory expectations, and stage of process maturity.
Unlock the power of your data
Turn complex bioprocess data into actionable knowledge with our team

